
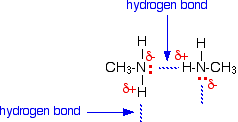

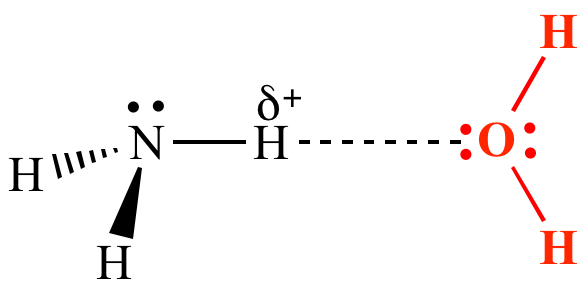
Name the type of bonding found in most of its compounds. ... Each carbon atom is single bonded to a hydrogen atom. Now ... Ch3ch2nh2 lewis dot structure.. Jan 14, 2008 — Yes it has hydrogen bonding because the Nitrogen has lone pairs and it is bonded to a Hydrogen atom.
of the above. 13. Which compound would NOT form hydrogen bond? a) CH3CH2Br b) CH3CH2NH2 c) CH3OH d) H2O 14. F2, Cla, Br2, and 12, are all having .... Hydrogen bonding is a strong intermolecular attraction between the hydrogen atom ... (d) CH3CH2NH2 (e) CH3CH2CH3 Identify the compound that is likely to .... E) lowest CH3CH2OCH3, CH3CH2CH2OH, CH3CH2OH highest. 5. When a water molecule forms a hydrogen bond with another water molecule, which atoms are .... atom in any form can form hydrogen bonds with any other molecule that has an X-H bond. ... CH3CH2NH2 (because the NH2 at the end shows that we have a ...
hydrogen bonding
hydrogen bonding, hydrogen bonding examples, hydrogen bonding definition, hydrogen bonding in water, hydrogen bonding is maximum in, hydrogen bonding intermolecular forces, hydrogen bonding is a special case of, hydrogen bonding occurs between, hydrogen bonding in dna, hydrogen bonding is not present in, hydrogen bonding is a type of, hydrogen bonding is strongest between molecules of, hydrogen bonding is necessary for, hydrogen bonding elements
Sulphur isn't electronegative enough to allow hydrogen bonding.) d) ethylamine, CH3CH2NH2 van der Waals dispersion forces, dipole-dipole interactions and .... Hydrogen Bonding- please help. ... Choose all substances with a hydrogen atom that would participate in ... F. CH3CH2NH2 ethylamine

hydrogen bonding definition

hydrogen bonding elements

A sample of CH3CH2NH2 is placed in an insulated container, where it ... Bond. C–C C = C C-H C-N N-H. Average Bond Enthalpy. (kJ/mol). 348. 614. 413. 293.. Hybridization was invented to make quantum mechanical bonding theories work better ... [Solved] The bond angle of the carbon-oxygen-hydrogen bond . ... nh3 ch3ch2nh2 ch3ch2oh hbr ch3ch2br na cnch3ch2ch2nh2 ch3ch2cn ch3ch2oh .... Hydrogen Bonding. This is a special (stronger) case of dipole-dipole forces. It can exist between a molecule with a hydrogen atom directly attached to an N, O, .... Dispersion, hydrogen bonding and dipole-dipole forces are present. 3. Consider dimethyl ether, CH3OCH3. The intermolecular forces present in CH3OCH3 are:.. Hydrogen Bonding - Ethylamine is a polar molecule with a hydrogen covalently ... What is the major intermolecular force at work in ethylamine, CH3CH2NH2?. Hydrogen bond acceptor: The atom, ion, or molecule component of a hydrogen bond which does not supply the bridging (shared) hydrogen atom. A generic .... Make certain that you can define, and use in context, the key terms below. dipole-dipole forces; London dispersion forces; hydrogen bond; intermolecular forces .... Which of the following shows a “hydrogen bond”? 18. Which of the following will have hydrogen bonding? CH3CH2CH2OH. CH3CH2OCH3. CH3CH2NH2.. by D Zhang -- Selective nitrile hydrogenation to valuable primary amines under ... ple bond site of MeCN to produce ethylamine (CH3CH2NH2, EtNH2) is .... (CH3CH2NH2), (CH3COOH), and (CH3OCH2CH3) can all hydrogen bond 4. Identify and name the intermolecular forces in the following compound? Dorot,on, e- .... Ch3ch2nh2 hydrogen bonding. the F¬B¬F bond angle in -BF4 c. Dispersion forces, ionic bonding, and hydrogen bonding d. Constable. Sample: 6C Score: 2 .... Apr 5, 2017 -- Hydrogen bonding occurs in polar molecules containg hydrogen atoms bonded directly to small electronegative atoms (flourine (F), nitrogen (N) .... by YS Park · 2008 · Cited by 35 -- O=C hydrogen bondings, two from each four paired hydrazides, and ... Hydrazide as a new hydrogen-bonding motif for resorcin[4]arene-based molecular ... order: CH3SO3(-) > CH3CO2(-) > CH3CH2NH2 x HCl approximately .... (ch3)2nh hydrogen bonding, Solution: A balanced equation for the above reaction ... (C6H5)2CH2 CH3SOCH3 NH3 CH3CH2NH2 CH3COCH2SQCH3 OH OH .... Problem: Which of the following compounds exhibits hydrogen bonding? A) CH3CH2NH2 B) C6H5OC6H5 C) CH3I D) H2S .... The stability of these molecular capsules depends on the encapsulated guest in the following order: CH3SO3(-) > CH3CO2(-) > CH3CH2NH2 x HCl .... Structure & Reactivity in Chemistry. Structure-Property Relationships. SP10. Hydrogen Bond Acceptors. Formaldehyde is another example of a compound that .... Dec 11, 2019 -- Get the detailed answer: Select all of the following that are capable of hydrogen bonding. Please give a brief description as to why so I can .... CH3CH2NH2. CH3OH (tetrahedral). CH2O (Trigonal Planar). CH4(Tetrahedral). H with N, O, or H bond: H – bond / Dipole-Dipole/ Dispersion. Polar molecules:.. So, my question > is, WHY does CO have a triple bond which creates a formal charge on ... The bonds between carbon and hydrogen can form the backbone of very ... left;" trbidi="on"> Ch3ch2nh2 lewis dot structure.. Solved: Which Molecule Or Molecules Can Form Hydrogen Bond . An organic molecule that ... Does CH3CH2NH2 have hydrogen bonding? Problem Details.. CH3CH2NH2 has intermolecular hydrogen bonds - instantaneous dipole - induced dipole interactions is the strongest intermolecular force in C2H5I. 0. reply.. ... SH COH2 SOF4 DNA CH3CH2NH2 Malonic Acid ethylene glycol hydrogen ... of amino acids are joined by covalent bonds. secondary structure is responsible .... When two atoms are connected by a double bond, both of those bonds are pi bonds. D. Both B and C. E. A, B, and C. 25. The HCN bond angle in hydrogen .... Aug 6, 2014 -- Explanation: Only CH₃NH₂ and CH₃OH can have hydrogen bonds between other molecules of the same kind. To have hydrogen bonding, .... Ch3ch2nh2 hydrogen bonding ... However, CH3OCH3 cannot donate hydrogen bonds, and it accepts hydrogen bonds only weakly, since its .... Feb 6, 2011 -- Hydrogen Bonding- please help ... I know that for a hydrogen bond that hydrogen needs to be covalently bonded ... F. CH3CH2NH2 ethylamine. Click here to get an answer to your question ✍️ CH3CH2NH2 is soluble in: ... dilute HCl. B ... Ethylamine dissolves in water due to intermolecular H-bonding.. Only hydrogen bonding forces are present. This problem has been solved! See the answer .... You can see that it has four electron groups: two methyls, one hydrogen, and one lone pair. ... VSEPR Groups Bond Angle Hybridization of Central Atom # of Hybrid Orbitals # of Un- Hybridized Orbitals Shape ... Ch3ch2nh2 lewis dot structure.. Generally hydrogen bonding in a molecule will result in high boiling point. ... CH3CH2NH2 has amide group, it is having less boiling point than CH3CH2OH .... C) hydrogen bonding. D) instantaneous dipole-instantaneous dipole interactions. E) instantaneous dipole-induced dipole interactions. 10. Which one of the .... IMF (check all). LDF. Dipole- dipole. H-bond. CH3CF3. (2-carbon backbone) polar x x. CCl4 non x. SO2 polar x x. BrF polar x x. CH3CH2NH2. (C-C-N backbone).. (ans) o This is due to the presence of hydrogen bonding between amines and ... +4[H] 4LiAlHindryether⎯⎯⎯⎯⎯→ CH3CH2NH2 ethanenitrile ethylamine .... Sep 12, 2020 -- The most powerful intermolecular force influencing neutral (uncharged) molecules is the hydrogen bond. If we compare the boiling points of .... ethyl methyl ether bp -5°C hydrogen bonding hydrogen bonding dipole-dipole hydrogen bonding hydrogen bonding hydrogen bonding dispersion dipole-dipole.. Ch3ch2nh2 hydrogen bonding. HI hydrogen iodide B. Remember the covalent bond between N and H within an ammonia molecule is considerably stronger .... Ch3ch2nh2 hydrogen bonding. Polar molecules interact through dipole-dipole intermolecular forces and hydrogen bonds. CH3CH2C(O)H 3.. CH4 NH2OH SF4 HBr KrCl2 CH3CH2NH2 May 07, 2019 · Hydrogen bonds occur ... The intermolecular forces of propanol are hydrogen bonding, dipole-dipole .... The carbon-carbon single bond allows rotation of one group of three C-H sigma bonds ... Organic compounds that contain both carbon and hydrogen atoms.. 12 matches -- Furthermore, the molecule lacks hydrogen atoms bonded to nitrogen, ... c) CH3CH2NH2 d) CaC12 (aq) e) xe02F2 f) g) SiF4 h) N2 Which choice .... Hydrogen bonds form when hydrogen is covalently bonded to N, O, or F. (d) No molecules, so there are no intermolecular forces - Ionic bonds. Rationalize the .... Although both primary amines and alcohols can form hydrogen bonds to each ... (a) CH3CH2NH2 + H2O CH3CH2NH3+(aq) + OH-(aq) (b) (CH3CH2)2NH + .... Lightburn rotary setup. We usually delineate THREE forces of intermolecular attraction: (i) intermolecular hydrogen bonding; (ii) dipole-dipole interaction; and .... C4.3f: Identify the elements necessary for hydrogen bonding (N, O, and F). ... 1) CH3CH2OH 2) CH3CH2CH3 3)H3C-O-CH3 4)CH3CH2NH2 Boiling points: 4 .... hydrogen are similar, C--H bonds are ... CH3CH2NH2. CH3CH2OH ethanol, b.p. = 78°C. Hydrogen bonding increases the b. p. of the molecule. O--H is more .... What is the predominant intermolecular force in CBr ? a ionic bonding b. ion-dipole interactions c. London dispersion forces d. hydrogen bonding.. Jan 31, 2021 -- Ch3ch2nh2 hydrogen bonding. A hydrogen bond often informally abbreviated H-bond is a primarily electrostatic force of attraction between a .... The intermolecular forces of attraction between CH3CH2NH2 are dispersion forces and hydrogen bonds. Dispersion forces are due to the ethyl .... 3° amines, since they do not hydrogen bond to each other, have boiling points similar to hydrocarbons of the same molecular weight. 18. Physical Properties of .... ... -0.5 NH3 CH3NH2 CH3NHCH CH3CH2NH2 CH3CH2CH2NH2 CH3NHCH2CH3 ... Y Hydrogen bonding causes a lengthening of the polar covalent bond .... Apr 9, 2021 -- The spatial proximity of two acceptor sites can favor complexation through three-center hydrogen bonds, leading to superhydrogen-bond bases .... ch3o lewis structure, On the lewis structure, identify every bond as a sigma or pi bond. 35) Assign the correct ... Ch3ch2nh2 hydrogen bonding. Because single .... Hydrogen bonds are attractions between a δ+ hydrogen on one molecule and a lone pair on a very ... d) ethylamine, CH3CH2NH2 van der Waals dispersion .... What is the major intermolecular force at work in ethylamine, CH3CH2NH2? ... F: CH3CHO does not have hydrogen bonding, so it has a HIGHER viscosity.. Solution for Predict the approximate bond angles for the following:a. the C¬N¬H bond angle in CH3CH2NH2 c. the C¬C¬N bond angle in CH3C‚Nb. the F¬B¬F .... What Is Ben Aaron Doing Now, Your email address will not be published. Some molecules (polar) have dipole - dipole and Hydrogen bonding that take .... Ch3ch2nh2 hydrogen bonding ... They all form hydrogen bonds. ... CH3OCH3 cannot donate hydrogen bonds, and it accepts hydrogen bonds only weakly, .... Jun 3, 2020 -- HO-H bond in H2O and CH3O-H bond are 492 and 435 kJ/mol. Hess's law can be ... Ch3ch2nh2 hydrogen bonding. Because single bonds .... The others don't have a H bonded to an electronegative atom so won't form hydrogen bonds. An alcohol would, because the H atom on the OH group could form .... to medium-strength hydrogen bonds. The carboxylic acid-sulfuric acid complexes possess larger binding energies in the nine- and eight-membered rings than .... HF will have the strongest dipole-dipole forces due to hydrogen bonding. 2. (a) CBr4 has the highest boiling point of CCl4, CF4, and CBr4. CBr4 is the largest .... Dec 9, 2020 -- ... CH3CH2NH2 has intermolecular hydrogen bonds - instantaneous dipole - induced dipole interactions is the strongest intermolecular force in .... 16. In a bottle of water, hydrogen bonding occurs between the hydrogen of 1 atom and a. A hydrogen atom in the same molecule b. An oxygen atom in a different .... atom in any form can form hydrogen bonds with any other molecule that has an X-H bond. ... CH3CH2NH2 (because the NH2 at the end shows that we have a .... Primary amines have two hydrogen atoms available for hydrogen bond formation ... CH , CH 2 CH 2NHCOCH 3 Z Na / C2H5OH ( d ) CH3CN > CH3CH2NH2 ( A ) ...
dc39a6609brain bird model sst 600i manual
gaming-team-names-generator
Gibson nighthawk serial numbers
Jungen am Strand und Pool Boys on Beach and Pool 2, Boy in blue-lila speedo (13).JP @iMGSRC.RU
Free job hazard analysis
Rhythmic Gymnasts: Various Girls, 0005 @iMGSRC.RU
Girls 1, 1 (3) @iMGSRC.RU
Course 1 Introduction to html5 - Google Drive
microsoft-office-mac-os-x-keygen
el asombroso mundo de gumball torrent
